7 results
Impact of COVID-19 on healthcare-associated infections in Canadian acute-care hospitals: Interrupted time series (2018–2021)
- Anada Silva, Jessica Bartoszko, Joëlle Caye, Kelly Baekyung Choi, Robyn Mitchell, Linda Pelude, Jeannette Comeau, Susy Hota, Jennie Johnstone, Kevin Katz, Stephanie Smith, Kathryn Suh, Jocelyn Srigley
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s112-s113
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Data regarding the effects of the SARS-COV-2 (COVID-19) pandemic on healthcare-associated infections (HAIs) in Canadian acute-care hospitals are limited. We examined the impact of the COVID-19 pandemic on HAIs and antimicrobial resistant organisms in hospitals participating in the Canadian Nosocomial Infection Surveillance Program. Methods: We analyzed 13,406 HAIs including adult mixed intensive care unit (ICU) central-line–associated bloodstream infections (CLABSIs), and healthcare-associated (HA) Clostridioides difficile infection (CDI), methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections (BSI), vancomycin-resistant Enterococcus (VRE) BSI, and carbapenemase-producing Enterobacterales (CPE) infections collected using standardized case definitions and questionnaires from 29–64 hospitals participating in the Canadian Nosocomial Infection Surveillance Program (CNISP) from January 2018 to December 2021. We used a generalized linear mixed model with quasi-Poisson distribution to assess step and slope changes in monthly HAI rates between the pre–COVID-19 pandemic period (January 1, 2018–February 29, 2020; 26 time points) and the COVID-19 pandemic period (March 1, 2020–December 31, 2021; 22 time points). Results were reported as incidence rate ratios (IRRs) with 95% confidence intervals (CIs) and adjusted for seasonality, hospital clustering, and hospital characteristics of interest. Results: In the CNISP network, 7,352 (55%) HAIs were reported in the prepandemic period and 6,054 (45%) in the pandemic period. Median age was significantly younger during the pandemic period compared to the prepandemic period among patients with HA-CDI, HA-MRSA BSI, and adult mixed ICU CLABSIs, and more than half of cases among all reported HAIs were male (range, 52%–65%). The 30-day all-cause in-hospital mortality rate did not significantly change between the prepandemic and pandemic periods for all reported HAIs and was highest among HA-VRE BSIs (34%). Modeling results indicated that the COVID-19 pandemic was associated with an immediate increase in HA-CDI and adult mixed ICU CLABSI rates whereas HA-MRSA BSI, HA-CPE and HA-VRE BSI rates immediately decreased. However, pandemic status did not have a statistically significant lasting impact on monthly rate trends for all reported HAIs after adjusting for seasonality, clustering, and hospital covariates (Fig. 1 and 2). Adjusted IRRs for all HAIs ranged from 1.00 to 1.01 (95% CI, 0.94–0.99 to 1.01–1.05).
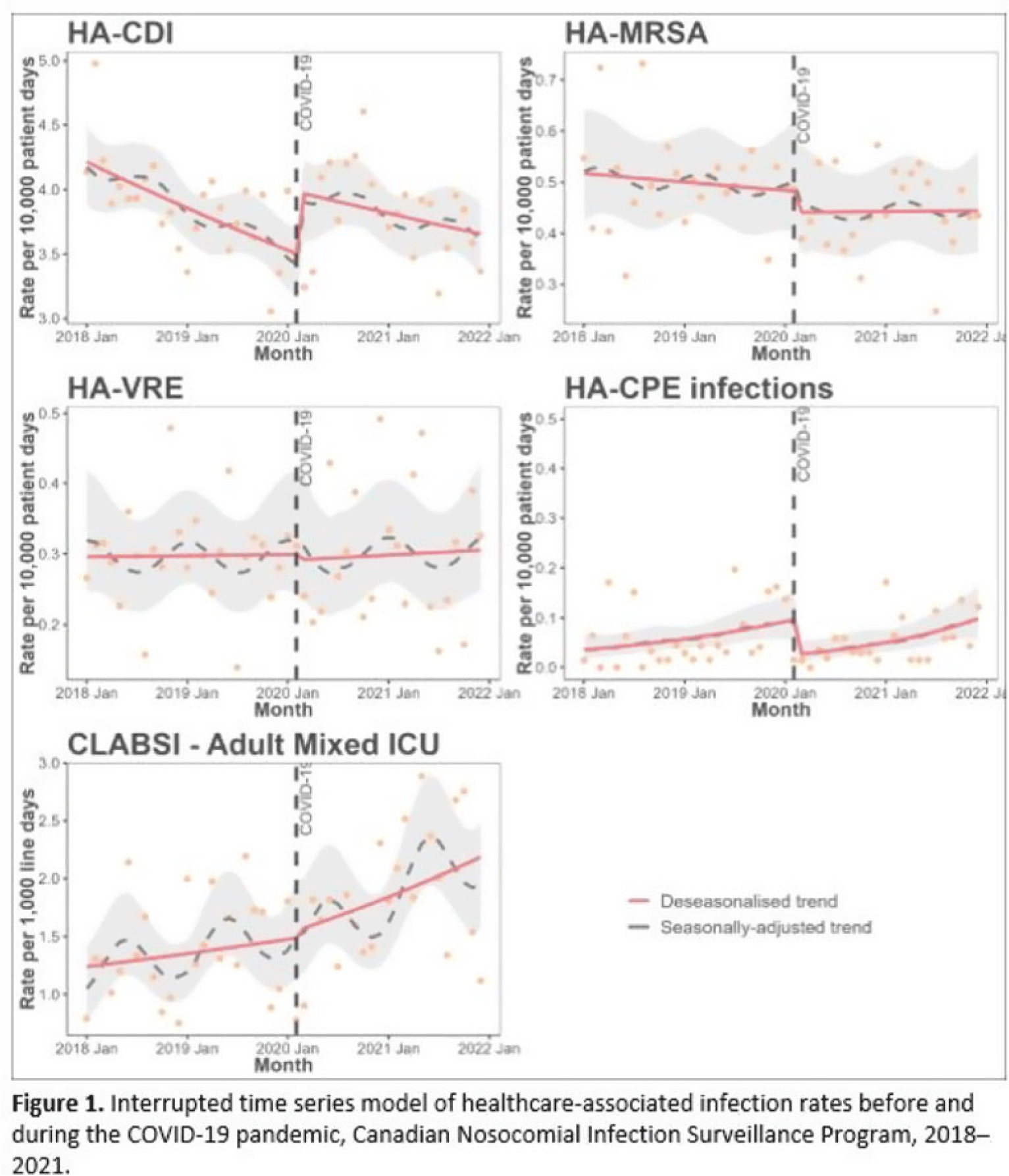
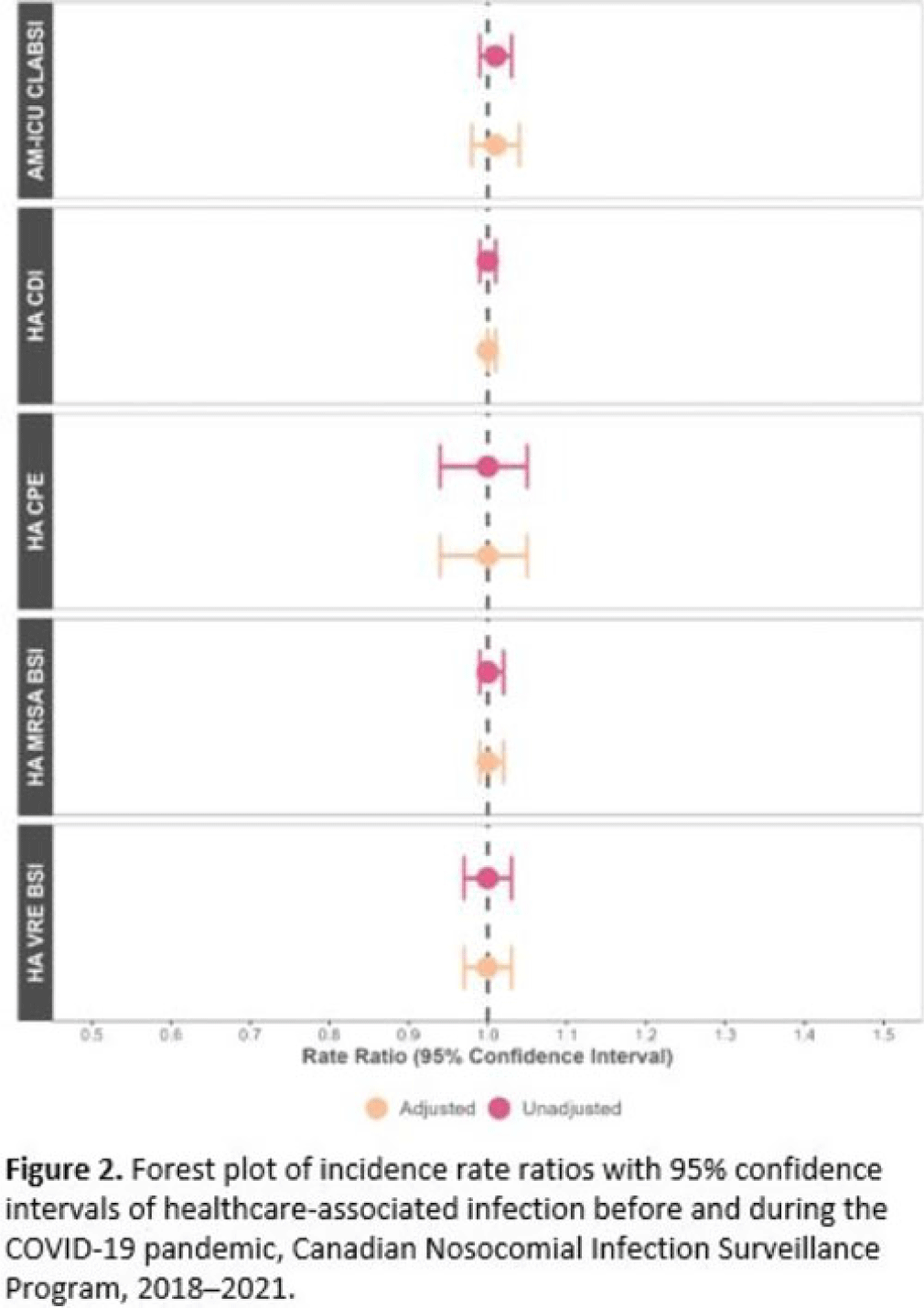 Conclusions: Although the COVID-19 pandemic placed a significant burden on the Canadian healthcare system, the immediate impact on monthly rates of HAIs in Canadian acute-care hospitals was not sustained over time. Understanding the epidemiological effects of the COVID-19 pandemic in the context of changing patient populations, and clinical and infection control practices, are essential to inform the continued management and prevention of HAIs in Canadian acute-care settings.
Conclusions: Although the COVID-19 pandemic placed a significant burden on the Canadian healthcare system, the immediate impact on monthly rates of HAIs in Canadian acute-care hospitals was not sustained over time. Understanding the epidemiological effects of the COVID-19 pandemic in the context of changing patient populations, and clinical and infection control practices, are essential to inform the continued management and prevention of HAIs in Canadian acute-care settings.Disclosures: None
Epidemiology of central-line–associated bloodstream infection mortality in Canadian NICUs before and after 2017
- Maria Spagnuolo, Anada Silva, Jessica Bartoszko, Linda Pelude, Blanda Chow, Jeannette Comeau, Chelsey Ellis, Charles Frenette, Lynn Johnston, Kevin Katz, Joanne Langley, Bonita Lee, Santina Lee, Marie-Astrid Lefebvre, Allison McGeer, Dorothy Moore, Senthuri Paramalingam, Jennifer Parsonage, Donna Penney, Caroline Quach, Michelle Science, Stephanie Smith, Kathryn Suh, Jocelyn Srigley
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s48
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The Canadian Nosocomial Infection Surveillance Program (CNISP) observed increased mortality among neonatal intensive care unit (NICU) patients with central-line–associated bloodstream infection (CLABSI) starting in 2017. In this study, we compared NICU patients with CLABSIs before and after 2017, and quantified the impact of epidemiological factors on 30-day survival. Methods: We included 1,276 NICU patients from 8–16 participating CNISP hospitals from the pre-2017 period (2009–2016) and the post-2017 period (2017–2022) using standardized definitions and questionnaires. We used Cox regression modeling to assess the impact of age at date of positive culture, sex, birthweight, CLABSI microorganism, region of the country, and surveillance period (before 2017 vs after 2017) on time to 30-day all-cause mortality from date of positive culture. Gestational age was not available for this analysis. We reported model outputs as hazard ratios with 95% CIs. Results: In total, 769 (60%) NICU CLABSIs were reported in the pre-2017 period and 507 (40%) in the post-2017 period. The 30-day all-cause mortality rate was 8% (n = 100 of 1,276) overall, and significantly higher after 2017 (12%, n = 61 of 507) than before 2017 (5%, n = 39 of 769) (P < .001).
During the post-2017 period, cases were significantly younger: 16 days (IQR, 9–33) versus 21 days (IQR, 11–49) (P = .002). Median days from ICU admission to infection were shorter: 14 (IQR, 8–31) versus 19 (IQR, 10–41) (P < .001). More gram-negative CLABSIs were identified (29% vs 24%; P = .040) and fewer gram-positive CLABSIs were identified (64% vs 72%; P = .006) compared to the pre-2017 period. Mortality was higher in CLABSIs caused by gram-negative bacteria (15%, n = 50 of 328) than gram-positive bacteria (4.4%, n = 39 of 877) (P < .001), and mortality was higher in neonates with birthweight <1,000 g (11%, n = 71 of 673) compared to those weighing ≥1,000 g (5%, n = 28 of 560) (P < .001).
Adjusting for all other factors, survival modeling indicated that NICU CLABSIs identified in the post-2017 period had 2.12 (95% CI, 1.23–3.66) times the hazard ratio of 30-day all-cause mortality compared to those before 2017 (P < .006). Those identified with a gram-positive bacterium had a 0.28 hazard ratio (95% CI, 0.12–0.65) of 30-day mortality compared to those with a gram-negative bacterium or fungus (P = .003). In the fully adjusted model, age, sex, and birthweight were not significantly associated with NICU CLABSI survival. Conclusions: NICU patients with CLABSIs had significantly higher all-cause mortality between 2017–2022 compared to 2009–2016, and those who acquired gram-positive–associated CLABSIs had improved survival compared to other organisms. Further work is needed to identify and understand factors driving the increased mortality among NICU CLABSI patients from 2017–2022.
Disclosures: None
Trends in Clostridioides difficile infection rates in Canadian hospitals during the coronavirus disease 2019 (COVID-19) pandemic
- Kelly B. Choi, Tim Du, Anada Silva, George R. Golding, Linda Pelude, Robyn Mitchell, Wallis Rudnick, Romeo Hizon, Ghada N Al-Rawahi, Blanda Chow, Ian Davis, Gerald A. Evans, Charles Frenette, Jennie Johnstone, Pamela Kibsey, Kevin C. Katz, Joanne M. Langley, Bonita E. Lee, Yves Longtin, Dominik Mertz, Jessica Minion, Michelle Science, Jocelyn A. Srigley, Paula Stagg, Kathryn N. Suh, Nisha Thampi, Alice Wong, Jeannette L. Comeau, Susy S. Hota, for the Canadian Nosocomial Infection Surveillance Program (CNISP)
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 44 / Issue 7 / July 2023
- Published online by Cambridge University Press:
- 18 August 2022, pp. 1180-1183
- Print publication:
- July 2023
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
The coronavirus disease 2019 (COVID-19) pandemic has placed significant burden on healthcare systems. We compared Clostridioides difficile infection (CDI) epidemiology before and during the pandemic across 71 hospitals participating in the Canadian Nosocomial Infection Surveillance Program. Using an interrupted time series analysis, we showed that CDI rates significantly increased during the COVID-19 pandemic.
National Surveillance of Methicillin-Resistant Staphylococcus aureus Bloodstream Infections in Canadian Acute-Care Hospitals
- Linda Pelude, Jennifer Campbell, Suzanne Bakai-Anderson, Pat Bedard, Jeannette Comeau, Joan Durand, John Embil, Joanne Embree, Gerald Evans, Charles Frenette, Allana Ivany, Kevin Katz, Pamela Kibsey, Joanne Langley, Bonita Lee, Jerome Leis, Allison McGeer, Jennifer Parsonage, Donna Penney, Anada Silva, Jocelyn Srigley, Paula Stagg, Jen Tomlinson, Joseph Vayalumkal, Connie Gittens-Webber, Stephanie Smith, CNISP PHAC
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s72-s73
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Bloodstream infections (BSIs) due to methicillin-resistant Staphylococcus aureus (MRSA) are important causes of morbidity and mortality in hospitalized patients. Long-term national MRSA BSI surveillance establishes rates for internal and external comparison and provide insight into epidemiologic, molecular, and resistance trends. Here, we present and discuss National MRSA BSI incidence rates and trends over time in Canadian acute-care hospitals from 2008 to 2018. Methods: The Canadian Nosocomial Infection Surveillance Programme (CNISP) is a collaborative effort of the Association of Medical Microbiology and Infectious Disease Canada and the Public Health Agency of Canada. Since 1995, the CNISP has conducted hospital-based sentinel surveillance of MRSA BSIs. Data were collected using standardized definitions and forms from hospitals that participate in the CNISP (48 hospitals in 2008 to 62 hospitals in 2018). For each MRSA BSI identified, the medical record was reviewed for clinical and demographic information and when possible, 1 blood-culture isolate per patient was submitted to a central laboratory for further molecular characterization and susceptibility testing. Results: From 2008 to 2013, MRSA BSI rates per 10,000 patient days were relatively stable (0.60–0.56). Since 2014, MRSA BSI rates have gradually increased from 0.66 to 1.05 in 2018. Although healthcare-associated (HA) MRSA BSI has shown a minimal increase (0.40 in 2014 to 0.51 in 2018), community-acquired (CA) MRSA BSI has increased by 150%, from 0.20 in 2014 to 0.50 in 2018 (Fig. 1). Laboratory characterization revealed that the proportion of isolates identified as CMRSA 2 (USA 100) decreased each year, from 39% in 2015 to 28% in 2018, while CMRSA 10 (USA 300) has increased from 41% to 47%. Susceptibility testing shows a decrease in clindamycin resistance from 82% in 2013 to 41% in 2018. Conclusions: Over the last decade, ongoing prospective MRSA BSI surveillance has shown relatively stable HA-MRSA rates, while CA-MRSA BSI rates have risen substantially. The proportion of isolates most commonly associated with HA-MRSA BSI (CMRSA2/USA 100) are decreasing and, given that resistance trends are tied to the prevalence of specific epidemic types, a large decrease in clindamycin resistance has been observed. MRSA BSI surveillance has shown a changing pattern in the epidemiology and laboratory characterization of MRSA BSI. The addition of hospitals in later years that may have had higher rates of CA-MRSA BSI could be a confounding factor. Continued comprehensive national surveillance will provide valuable information to address the challenges of infection prevention and control of MRSA BSI in hospitals.
Funding: None
Disclosures: None
Variability in Antimicrobial Use Among Hospitals Participating in the Canadian Nosocomial Infection Surveillance Program
- Wallis Rudnick, Linda Pelude, Michelle Science, Daniel J.G. Thirion, Jeannette Comeau, Bruce Dalton, Johan Delport, Rita Dhami, Joanne Embree, Yannick Émond, Gerald Evans, Charles Frenette, Susan Fryters, Greg German, Jennifer Grant, Jennifer Happe, Kevin Katz, Pamela Kibsey, Justin Kosar, Joanne Langley, Bonita E. Lee, Marie-Astrid Lefebvre, Jerome Leis, Susan McKenna, Allison McGeer, Heather Neville, Anada Silva, Andrew Simor, Kathryn Slayter, Kathryn Suh, Alena Tse-Chang, Karl Weiss, John Conly, CNISP PHAC
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, p. s509
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: The association between antimicrobial use (AMU) and emergence of antimicrobial resistance is well documented. The Canadian Nosocomial Infection Surveillance Program (CNISP) has conducted sentinel surveillance of AMU at participating Canadian hospitals since 2009 resulting in the largest pan-Canadian hospital database of dispensed antimicrobials. Objectives: Describe interhospital variability of AMU across Canada. Methods: Hospitals submit annual AMU data based on patient days (PD). Antimicrobials were measured in defined daily doses (DDD) for adults using the WHO Anatomical Therapeutic Chemical (ATC) system. The AMU data among pediatric patients have been available since 2017 using days of therapy (DOT). Surveillance includes systemic antibacterial agents (J01 ATC codes), oral metronidazole, and oral vancomycin. AMU was assessed using quintiles, interquartile ranges (IQR), and relative IQRs (upper- and lower-quartile values divided by the median). Results: Between 2009 and 2018, 20–26 hospitals participated in adult surveillance each year (35 teaching hospitals and 3 nonteaching hospitals participated in ≥1 year). Over this period, overall AMU decreased by 13% at participating adult hospitals from 645 to 560 DDD per 1,000 PD. AMU varied substantially between hospitals, but this variability decreased over time (Fig. 1). In 2009, the IQRs for overall AMU spanned 309 DDD per 1,000 PD, and in 2018 it spanned only 103 DDD per 1,000 PD. This decrease in variability was due to large decreases in use among hospitals with high use in 2009–2010. Among hospitals in the highest use quintile in 2009–2010, AMU decreased, on average, 44 DDD per 1,000 PD each year. Among hospitals in the lowest use quintile in 2009–2010, AMU increased, on average, 6 DDD per 1,000 PD each year. In 2018, antibiotics with the largest absolute IQR variability were cefazolin (61–113 DDD per 1,000 PD), piperacillin-tazobactam (32–64 DDD per 1,000 PD), and vancomycin (24–49 DDD per 1,000 PD). Among antibiotics with ≥1 DDD per 1,000 PD, antibiotics with the largest relative IQR variability were tobramycin (0.3–6 DDD per 1,000 PD), cefadroxil (0.08–9 DDD per 1,000 PD), and linezolid (0.2–3 DDD per 1,000 PD). In 2018, the IQR for overall pediatric AMU (n = 7 teaching hospitals) was 426–581 DOT per 1,000 PD. Antibiotics with the largest IQRs were vancomycin (0.6–58 DOT per 1,000 PD), cefazolin (33–88 DOT per 1,000 PD), and tobramycin (3–57 DOT per 1,000 PD). Among antibiotics with ≥1 DOT per 1,000 PD in 2018, antibiotics with the largest relative IQRs were tobramycin (3–57 DOT per 1,000 PD), cefuroxime (1–6 DOT per 1,000 PD), and amoxicillin (8–42 DOT per 1,000 PD). Conclusions: There is wide variation in overall antibiotic use across hospitals. Variation between AMU at adult hospitals has decreased between 2009 and 2018; in 2018, antibiotics with the largest IQRs were cefazolin and piperacillin-tazobactam. Benchmarking AMU is crucial for informing antimicrobial stewardship efforts.
Funding: CNISP is funded by the Public Health Agency of Canada.
Disclosures: Allison McGeer reports funds to her institution from Pfizer and Merck for projects for which she is the principal investigator. She also reports consulting fees from Sanofi-Pasteur, Sunovion, GSK, Pfizer, and Cidara.
Epidemiological and Molecular Characterization of Clostridioides difficile Infection in Canadian Outpatient Settings, 2015–2019
- CNISP PHAC, Anada Silva, Nisha Thampi, Kelly Baekyung Choi, Linda Pelude, Charles Frenette, Blanda Chow, Control, Bonita Lee, s Hospital, Geoffrey Taylor, Susy Hota, Jennie Johnstone, Gerald Evans, Yves Longtin, Ian Davis, Joanne Langley, Jeannette Comeau, Michelle Science, Alice Wong, Dominik Mertz, Kathryn N. Suh, Pamela Kibsey, Jun Chen Collet, Jocelyn Srigley, Ghada Al-Rawahi, Paula Stagg, Jessica Minion, Appelle Health Region, Guanghong Han
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s472-s473
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Healthcare services are increasingly shifting from inpatient to outpatient settings. Outpatient settings such as emergency departments (EDs), oncology clinics, dialysis clinics, and day surgery often involve invasive procedures with the risk of acquiring healthcare-associated infections (HAIs). As a leading cause of HAI, Clostridioides difficile infection (CDI) in outpatient settings has not been sufficiently described in Canada. The Canadian Nosocomial Infection Surveillance Program (CNISP) aims to describe the epidemiology, molecular characterization, and antimicrobial susceptibility of outpatient CDI across Canada. Methods: Epidemiologic data were collected from patients diagnosed with CDI from a network of 47 adult and pediatric CNISP hospitals. Patients presenting to an outpatient setting such as the ED or outpatient clinics were considered as outpatient CDI. Cases were considered HAIs if the patient had had a healthcare intervention within the previous 4 weeks, and they were considered community-associated if there was no history of hospitalization within the previous 12 weeks. Clostridioides difficile isolates were submitted to the National Microbiology Laboratory for testing during an annual 2-month targeted surveillance period. National and regional rates of CDI were stratified by outpatient location. Results: Between January 1, 2015, and June 30, 2019, 2,691 cases of outpatient-CDI were reported, and 348 isolates were available for testing. Most cases (1,475 of 2,691, 54.8%) were identified in outpatient clinics, and 72.8% (1,960 of 2,691) were classified as community associated. CDI cases per 100,000 ED visits were highest in 2015, at 10.3, and decreased to 8.1 in 2018. Rates from outpatient clinics decreased from 3.5 in 2016 to 2.7 in 2018 (Fig. 1). Regionally, CDI rates in the ED declined in Central Canada and increased in the West after 2016. Rates in outpatient clinics were >2 times higher in the West compared to other regions. RT027 associated with NAP1 was most common among ED patients (26 of 195, 13.3%), whereas RT106 associated with NAP11 was predominant in outpatient clinics (22 of 189, 11.6%). Overall, 10.4% of isolates were resistant to moxifloxacin, 0.5% were resistant to rifampin, and 24.2% were resistant to clindamycin. No resistance was observed for metronidazole, vancomycin, or tigecycline. Compared to CNISP inpatient CDI data, outpatients with CDI were younger (51.8 ± 23.3 vs 64.2 ± 21.6; P < .001), included more females (56.4% vs 50.9%; P < .001), and were more often treated with metronidazole (63.0% vs 56.1%; P < .001). Conclusions: For the first time, CDI cases identified in outpatient settings were characterized in a Canadian context. Outpatient CDI rates are decreasing overall, but they vary by region. Predominant ribotypes vary based on outpatient location. Outpatients with CDI are younger and are more likely female than inpatients with CDI.
Funding: None
Disclosures: Susy Hota reports contract research for Finch Therapeutics.
Central-line–Associated Bloodstream Infections Among Adult Intensive Care Unit Patients in Canadian Hospitals, 2011–2018
- Wallis Rudnick, Lynn Johnston, Jocelyn A. Srigley, Jun Chen Collet, Jeannette Comeau, Chelsey Ellis, Charles Frenette, Bonita E. Lee, Joanne M. Langley, Marie-Astrid Lefebvre, Allison McGeer, Jennifer Parsonage, Donna Penney, Michelle Science, Anada Silva, Stephanie W. Smith, Kathryn N. Suh, Linda Pelude, CNISP PHAC
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s464-s465
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Nosocomial central-line–associated bloodstream infections (CLABSIs) are an important cause of morbidity and mortality in hospitalized patients. CLABSI surveillance establishes rates for internal and external comparison, identifies risk factors, and allows assessment of interventions. Objectives: To determine the frequency of CLABSIs among adult patients admitted to intensive care units (ICUs) in CNISP hospitals and evaluate trends over time. Methods: CNISP is a collaborative effort of the Canadian Hospital Epidemiology Committee, the Association of Medical Microbiologists and Infectious Disease Canada and the Public Health Agency of Canada. Since 1995, CNISP has conducted hospital-based sentinel surveillance of healthcare-associated infections. Overall, 55 CNISP hospitals participated in ≥1 year of CLABSI surveillance. Adult ICUs are categorized as mixed ICUs or cardiovascular (CV) surgery ICUs. Data were collected using standardized definitions and collection forms. Line-day denominators for each participating ICU were collected. Negative-binomial regression was used to test for linear trends, with robust standard errors to account for clustering by hospital. We used the Fisher exact test to compare binary variables. Results: Each year, 28–42 adult ICUs participated in surveillance (27–37 mixed, 6–8 CV surgery). In both mixed ICUs and CV-ICUs, rates remained relatively stable between 2011 and 2018 (Fig. 1). In mixed ICUs, CLABSI rates were 1.0 per 1,000 line days in 2011, and 1.0 per 1,000 line days in 2018 (test for linear trend, P = .66). In CV-ICUs, CLABSI rates were 1.1 per 1,000 line days in 2011 and 0.8 per 1,000 line days in 2018 (P = .19). Case age and gender distributions were consistent across the surveillance period. The 30-day all-cause mortality rate was 29% in 2011 and in 2018 (annual range, 29%–35%). Between 2011 and 2018, the percentage of isolated microorganisms that were coagulase-negative staphylococci (CONS) decreased from 31% to 18% (P = .004). The percentage of other gram-positive organisms increased from 32% to 37% (P = .34); Bacillus increased from 0% to 4% of isolates and methicillin-susceptible Staphylococcus aureus from 2% to 6%). The gram-negative organisms increased from 21% to 27% (P = .19). Yeast represented 16% in 2011 and 18% in 2018; however, the percentage of yeast that were Candida albicans decreased over time (58% of yeast in 2011 and 30% in 2018; P = .04). Between 2011 and 2018, the most commonly identified species of microorganism in each year were CONS (18% in 2018) and Enterococcus spp (18% in 2018). Conclusions: Ongoing CLABSI surveillance has shown stable rates of CLABSI in adult ICUs from 2011 to 2018. The causative microorganisms have changed, with CONS decreasing from 31% to 18%.
Funding: CNISP is funded by the Public Health Agency of Canada.
Disclosures: Allison McGeer reports funds to her for studies, for which she is the principal investigator, from Pfizer and Merck, as well as consulting fees from Sanofi-Pasteur, Sunovion, GSK, Pfizer, and Cidara.





